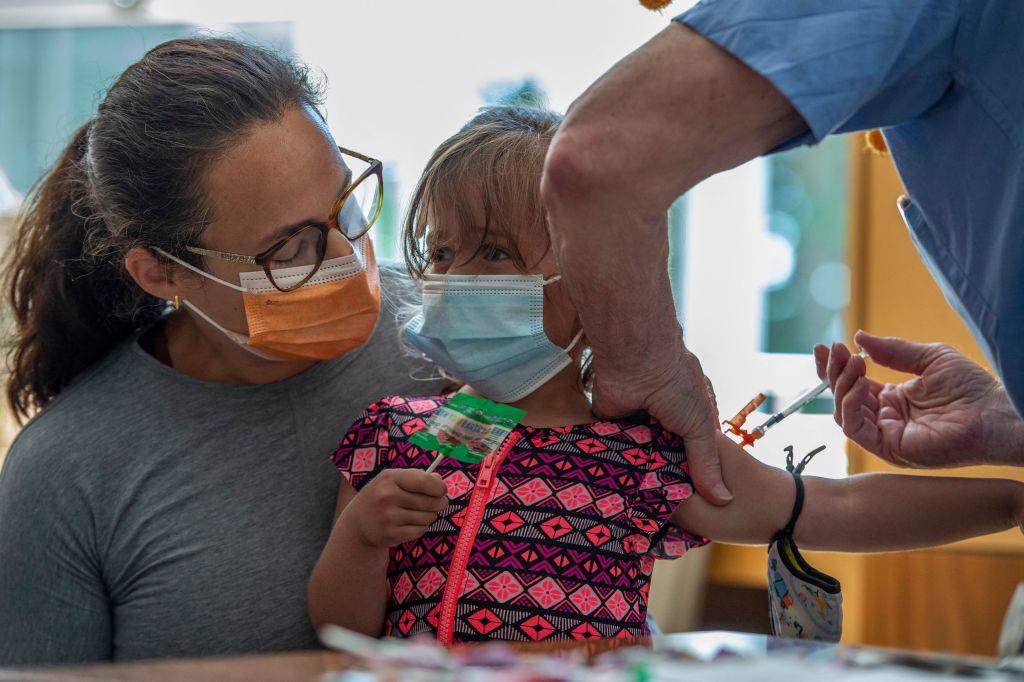
Pfizer BioNTech
-

CDC, Fauci Thinks COVID-19 Boosters Are Not Essential As of The Moment, to Address its Need in Briefing
-
Pfizer COVID-19 Booster to Get FDA Approval in August, Third Shot to Significantly Improve Immunity
-
mRNAs Are The Future of Vaccines After Pfizer, Moderna's Success Against COVID—HIV, Cancers Next-in-Line
-
FDA Approves Pfizer's COVID-19 Vaccine Emergency Use Despite Fears From Severe Allergic Reactions Reported in UK
Most Popular